Les programmes des chaires scientifiques d’excellence
Liryc a développé d’ambitieux programmes de recherche directement conduits par d’éminents scientifiques internationaux pour explorer des thématiques bien précises à fort potentiel d’innovation.
Chaire sur les mécanismes de la fibrillation atriale liés à l’âge
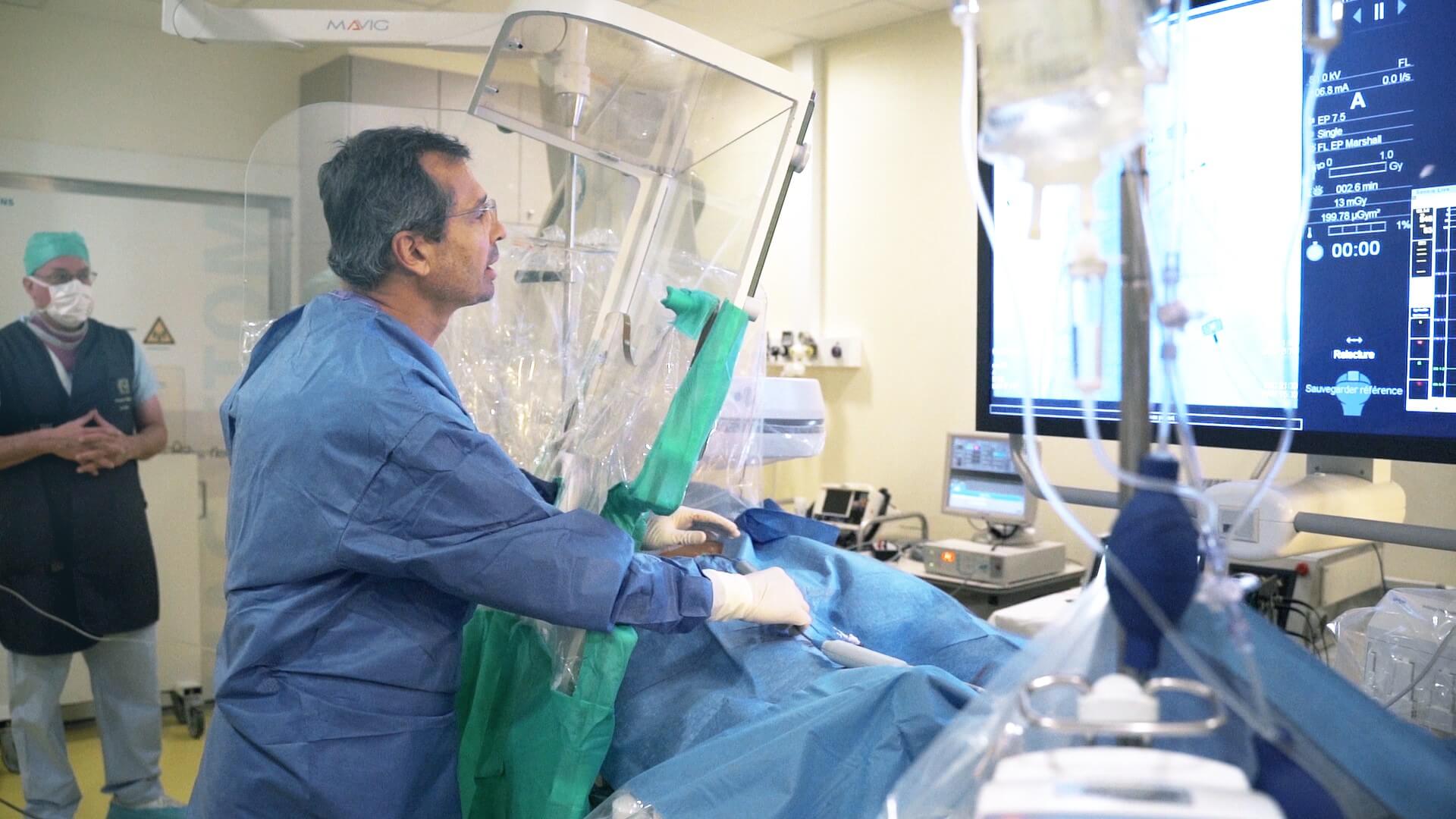
Evaluer les facteurs liés à l’âge de la fibrillation atriale
L’âge est identifié comme le facteur majoritairement responsable de l’apparition de la fibrillation atriale (FA). En effet, la prévalence de la FA augmente de façon exponentielle après 65 ans et elle est multipliée par 7 jusqu’à 85 ans. A contrario, les jeunes semblent être épargnés par la FA, puisque les personnes souffrant de cardiopathie congénitale et de troubles sévères au niveau des oreillettes développent rarement une FA avant l’âge de 45 ans. Néanmoins, la corrélation entre la FA et l’âge est mal comprise. Ce programme découle des travaux en cours à Liryc et d’études complémentaires menées à l’Institut de Cardiologie de Montréal (ICM) portant sur les facteurs contribuant au développement de la FA.
Objectifs du projet : Évaluer les facteurs liés à l’âge qui protègent le cœur des jeunes individus de la FA et rendent les individus plus âgés plus vulnérables.
Objectifs spécifiques :
(1) Description du remodelage électrique atriale métabolique et structurel chez de jeunes modèles expérimentaux versus des modèles âgés exposés à une FA.
(2) Identification de marqueurs pro/anti-FA, conception et test de nouveaux candidats thérapeutiques.
Le projet de cette chaire est réalisé en lien avec le programme ANR Unmasc. Les deux projets étudient la FA dans des modèles expérimentaux.
Améliorer les traitements de la fibrillation atriale
Ce programme devrait fournir de nouvelles informations majeures sur les mécanismes responsables de l’occurrence d’une FA. Les nouvelles connaissances obtenues amélioreront notre compréhension de l’arythmie et conduiront à l’identification de nouvelles cibles thérapeutiques. Ces cibles permettront ensuite d’améliorer les traitements ainsi que les stratégies pour optimiser la performance des approches thérapeutiques existantes comme l’ablation par cathéter.
Les collaborateurs principaux à Liryc
- Prof. Stanley Nattel, porteur de la chaire, Institut de Cardiologie de Montréal
- Dr Guido Caluori, post-doctorant, équipe Métabolisme à LIRYC
- Dr. Philippe Pasdois, maître de conférence, équipe Métabolisme à LIRYC
- Pr Pierre Jaïs, rythmologue au CHU de Bordeaux, professeur à l’Université de Bordeaux, directeur général de LIRYC
- Pr. Olivier Bernus, professeur, responsable du pôle physiopathologie de LIRYC
Chaire sur l’imagerie cardiaque
Améliorer la caractérisation détaillée du cœur
L’objectif est de développer une méthodologie d’IRM très avancée pour améliorer la caractérisation détaillée et quantitative du tissu cardiaque, de l’anatomie et de la fonction du myocarde. Il s’agit d’acquérir de nouvelles connaissances sur le substrat arythmogène améliorant ainsi la prise en charge des arythmies cardiaques.
Un autre objectif de cette chaire concerne l’éducation et la promotion de jeunes talents.
Pour atteindre ces objectifs, l’équipe s’appuie sur une méthodologie d’IRM reconnue, des équipements de pointe, des collaborations industrielles, une expertise de classe mondiale, un réseau international établi, de nouveaux concepts mathématiques et une collaboration interdisciplinaire et translationnelle.
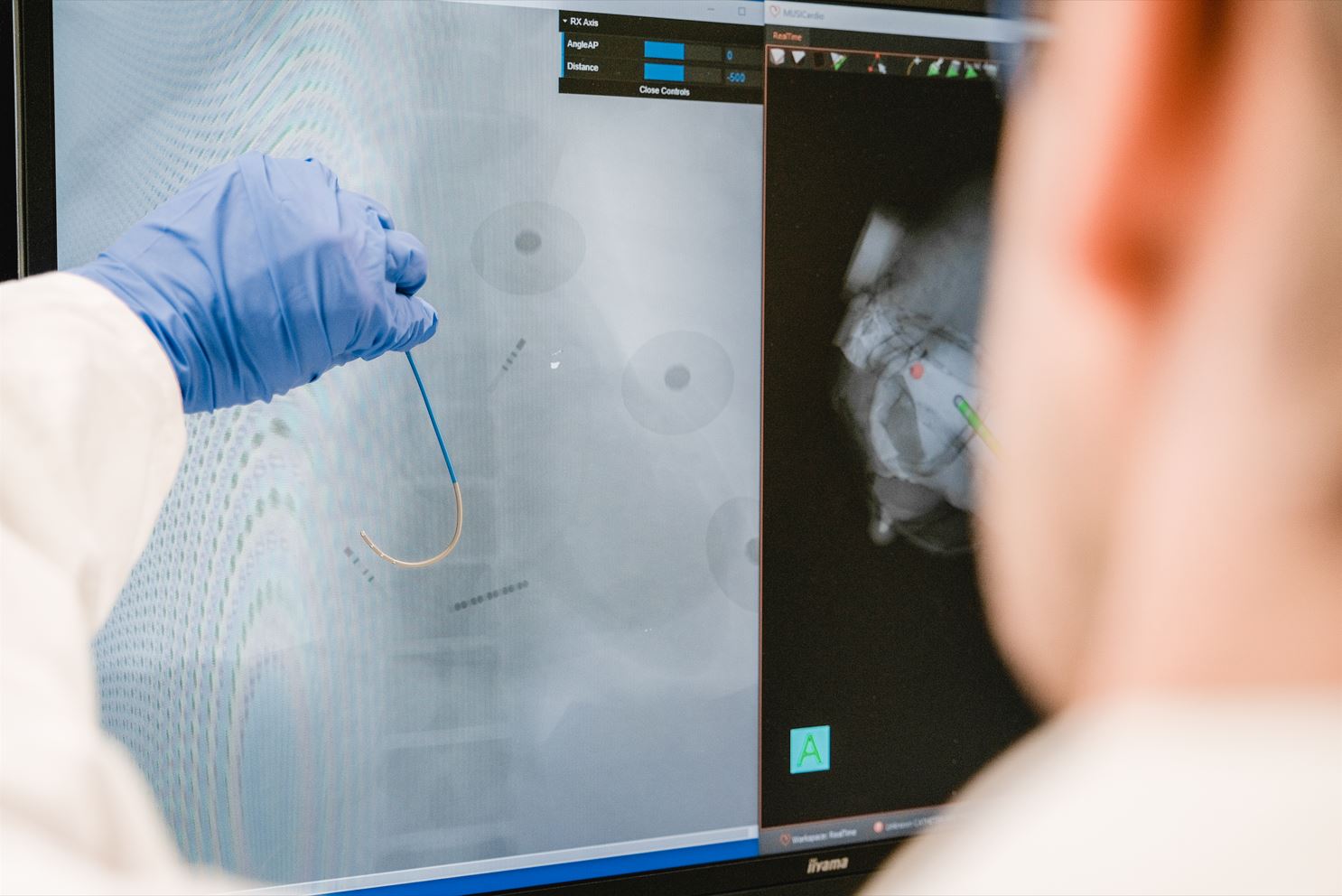
Un outil pour mieux accompagner les professionnels de santé
Ce programme devrait apporter de nouvelles connaissances dans le domaine des arythmies cardiaques ainsi qu’une technique de caractérisation non invasive, plus détaillée et quantitative du substrat myocardique. Elle permettra de mieux définir le succès ou l’échec des interventions électrophysiologiques et fournira un nouvel outil plus performant pour l’identification des patients susceptibles de bénéficier de la pose d’un défibrillateur cardiaque implantable (DCI).
Grâce à une approche rigoureusement interdisciplinaire et collaborative, nous nous appuyons sur l’expertise et l’esprit d’innovation des équipes pour de traduire les concepts mathématiques et physiques de l’IRM aux études d’imagerie fantôme, aux études in vivo, sur des volontaires sains, et finalement sur les patients en collaboration avec les professionnels de la santé.
Les collaborateurs principaux à Liryc
- Prof. Matthias Stuber de l’UNIL-CHUV de Lausanne, porteur de la chaire
- Aurélien Bustin, équipe du CHUV de Lausanne, post-doctorant, équipe imagerie à LIRYC, Centre de recherche en résonance magnétique cardiovasculaire (Lausanne, Suisse)
- Pr Hubert Cochet, cardiologue au CHU de Bordeaux, Professeur Université de Bordeaux, équipe d’imagerie médicale à LIRYC
- Pierre Jaïs, rythmologue au CHU de Bordeaux, Professeur Université de Bordeaux, Directeur Général de LIRYC
- Dr. Jérôme Yerly, docteur du CIBM-CHUV-UNIL
Chaire sur la neuromodulation par alcoolisation de la veine de Marshall
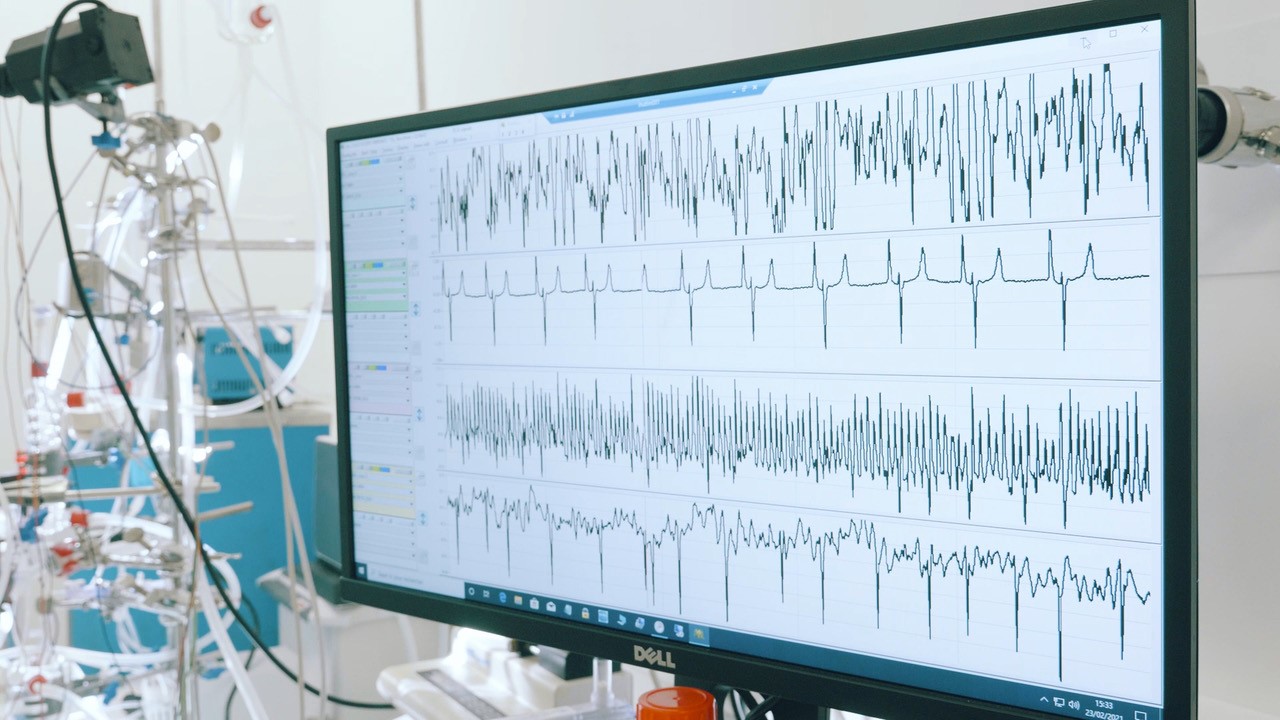
Etudier le rôle du système nerveux dans les arythmies
L’objectif principal de la chaire est d’étudier le rôle du système nerveux dans le déclenchement et la persistance des arythmies, ainsi que de comprendre comment l’ablation de l’innervation cardiaque peut protéger de ces arythmies.
(1) Nous avons développé une technique permettant d’enregistrer simultanément l’électrocardiogramme (ECG) et l’activité du nerf sympathique cutané (SKNA). Nous appelons cette technique la « neuECG ». Le premier objectif est de tester l’hypothèse selon laquelle l’injection d’alcool dans la veine de Marshall (VOM) peut réduire l’activité du nerf sympathique cutané chez les patients souffrant de fibrillation atriale (FA).
(2) Le second objectif est de tester l’hypothèse selon laquelle l’injection d’alcool dans les faisceaux nerveux sympathiques près de la veine cave supérieure gauche provoque un remodelage du ganglion stellaire.
Mieux prendre en charge les arythmies cardiaques
La neuromodulation est une méthode innovante prometteuse pour prendre en charge les patients souffrant d’arythmies cardiaques. Actuellement, l’équipe clinique de Liryc est leader dans le monde en ce qui concerne la méthode d’injection d’éthanol dans la VOM pour le traitement de la fibrillation atriale. Il est possible que cette même technique puisse être utilisée pour gérer la tachycardie ou la fibrillation ventriculaire. Si les résultats montrent que l’injection d’éthanol dans la VOM peut réduire avec succès le débit sympathique, nous réaliserons ensuite des études pour déterminer si cette technique peut être utilisée pour contrôler les arythmies ventriculaires et prévenir la mort subite cardiaque. Ces objectifs de recherche pourraient donc avoir un impact significatif majeur sur la pratique clinique.
Les collaborateurs principaux à Liryc
- Pr. Peng-Sheng Chen du Cedars-Sinai Medical Center à Los Angeles, porteur de la chaire
- Dr. Rokhaya Faye, PhD, équipe d’électrophysiologie tissulaire à LIRYC
- Dr. Meleze Hocini, MD, professeur associé au CHU de Bordeaux, directrice générale adjointe de LIRYC
- Pr. Olivier Bernus, professeur, responsable du pôle physiopathologie de LIRYC